Gamma-Stable Bio-Based Medical Trays Market Outlook 2026–2036: Global Market to Reach USD 1.7 Bn by 2036 at 6.57% CAGR
Japan’s orthopedic kit market is growing at a 5.1% CAGR, driven by high-precision manufacturing, material purity, and adoption of sustainable bio-based resins.
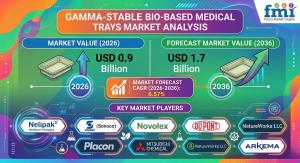
NEWARK, DE, UNITED STATES, March 17, 2026 /EINPresswire.com/ -- The global gamma-stable bio-based medical trays market is entering a phase of structural transformation as healthcare systems and medical device manufacturers accelerate the shift toward sustainable sterile barrier packaging. According to new analysis from Future Market Insights (FMI), the market was valued at USD 0.8 billion in 2025 and is projected to reach USD 0.9 billion in 2026, expanding at a compound annual growth rate (CAGR) of 6.57% between 2026 and 2036. By the end of the forecast period, the sector is expected to generate USD 1.7 billion in total market value.
The transition reflects growing demand for renewably sourced polymers capable of maintaining mechanical and sterile integrity after exposure to high-dose gamma radiation, a sterilization method widely used for medical devices. As hospital procurement standards evolve and sustainability metrics become embedded in purchasing decisions, medical packaging materials are being reassessed across the global healthcare supply chain.
Key Market Metrics
• Market Value (2026): USD 0.9 Billion
• Projected Market Value (2036): USD 1.7 Billion
• Forecast CAGR (2026–2036): 6.57%
• Leading Material Segment: Poly Lactic Acid (PLA)
• Top Application: Orthopedic Implant Packaging
• Dominant End-Use Sector: Hospitals
Discover Growth Opportunities in the Market – Get Your Sample Report Now
https://www.futuremarketinsights.com/reports/sample/rep-gb-32296
Market Overview: What Is Driving the Shift Toward Bio-Based Medical Trays?
Gamma-stable bio-based medical trays represent a specialized segment of the medical packaging industry. These trays are typically thermoformed or injection-molded rigid containers made from plant-derived polymers designed to maintain structural performance and microbial barrier integrity after exposure to ionizing radiation.
Unlike conventional fossil-based plastics such as PETG or high-impact polystyrene, bio-based trays are engineered using renewable feedstocks including polylactic acid (PLA), bio-polyethylene (PE), and bio-polypropylene (PP). These materials are formulated to withstand sterilization doses of 25–50 kilogray (kGy) while preserving dimensional stability, clarity, and impact resistance.
Healthcare institutions and device manufacturers are increasingly evaluating these materials not only for environmental benefits but also for operational performance across the sterile supply chain.
Procurement Pressure and Carbon Accountability Accelerate Adoption
Healthcare procurement strategies are evolving rapidly as hospital systems incorporate carbon accounting and sustainability metrics into supplier evaluations. For many medical device manufacturers, packaging is now a measurable component of Scope 3 emissions reporting.
Industry analysts note that the shift toward bio-based trays is not merely a substitution of materials but a broader reassessment of the sterile packaging lifecycle.
Quality assurance teams increasingly focus on whether renewable polymers can maintain impact resistance, dimensional stability, and seal integrity after gamma sterilization. Delaying adoption may expose manufacturers to procurement risks as hospitals prioritize vendors capable of providing lifecycle assessment data and transparent environmental reporting.
Key Demand Drivers Reshaping the Market
Several structural factors are influencing the adoption of gamma-stable bio-based medical trays across global healthcare systems.
Primary Demand Drivers
• Sustainability procurement mandates: Global hospital networks increasingly require suppliers to reduce the carbon footprint of single-use surgical packaging.
• Material science advancements: New bio-resin formulations now achieve performance levels comparable to traditional PETG in terms of clarity and impact strength.
• Regulatory pressure: Restrictions on certain fossil-based additives are encouraging medical device manufacturers to qualify renewable polymer alternatives.
• Sterile packaging innovation: High-modulus bio-polymers allow more compact tray designs, reducing packaging volume and logistics costs.
Segment Analysis: PLA Dominates Material Adoption
Within the material landscape, Poly Lactic Acid (PLA) is expected to hold 41.2% market share in 2026, making it the most widely adopted renewable polymer in the sector.
PLA provides several operational advantages for medical packaging converters:
• High optical clarity suitable for pre-surgical inspection
• Compatibility with existing thermoforming equipment
• Established pathway toward USP Class VI medical compliance
• Reliable wall thickness during vacuum forming processes
These attributes make PLA particularly attractive for device manufacturers transitioning from fossil-based plastics without requiring large capital investments in new manufacturing infrastructure.
Orthopedic Implants Lead Application Demand
The orthopedic implants segment represents the largest application category, projected to account for 34.6% of market share in 2026.
Orthopedic devices often feature complex geometries and sharp edges, requiring rigid trays with strong structural properties. Bio-based materials that maintain stiffness and dimensional stability under gamma irradiation allow manufacturers to safely package heavy surgical components while maintaining sterile barrier integrity.
Industry observers note that adoption often follows a phased pattern:
• Initial qualification: Tier-1 orthopedic OEMs conduct multi-year shelf-life validation studies.
• Standardization phase: Cardiovascular and surgical instrument manufacturers adopt validated materials.
• Mass conversion: Dental and single-use tool manufacturers convert once cost parity is achieved.
Hospitals Drive End-Use Adoption
Hospitals account for the largest end-use share at 48.2%, reflecting their role as the final decision-makers in the medical supply chain.
Procurement departments are increasingly evaluating packaging materials based on environmental impact as well as clinical performance. In many hospital systems, sustainability scoring is now included in request-for-proposal evaluations for surgical device suppliers.
Operational initiatives within hospitals include:
• Clinical validation testing to ensure trays do not generate particulates during aseptic opening
• Waste-stream segregation training for identifying compostable or recyclable bio-based materials
• ESG reporting integration linking packaging choices with environmental targets
Once validated for a single surgical product line, hospital networks frequently expand bio-based tray requirements across multiple device categories.
Regional Outlook: Asia Emerging as a Manufacturing Hub
Regional adoption patterns reflect differences in regulatory environments, healthcare procurement models, and manufacturing infrastructure.
Country-Level Growth Outlook (CAGR 2026–2036)
• India: 9.4%
• China: 8.8%
• United States: 7.2%
• Brazil: 6.9%
• Germany: 6.4%
• United Kingdom: 5.8%
• Japan: 5.1%
India and China are expected to lead global growth as both nations expand domestic medical device manufacturing capacity and invest in localized bioplastic production.
In India, government-supported MedTech parks are facilitating lower-cost manufacturing of sustainable medical packaging, positioning the country as a potential export hub for bio-based sterile trays.
Meanwhile, European adoption is strongly influenced by regulatory frameworks such as the Packaging and Packaging Waste Regulation (PPWR), which places increasing financial pressure on high-carbon plastic packaging.
Competitive Landscape and Industry Participants
The gamma-stable bio-based medical trays market is moderately concentrated due to the high regulatory and technical requirements involved in producing medical-grade sterile packaging.
Key companies operating in the sector include:
• Nelipak Healthcare Packaging
• TEQ (Sonoco)
• Novolex
• Placon
• DuPont
• Mitsubishi Chemical
• NatureWorks LLC
• Arkema
• Oliver Healthcare Packaging
• Steripack Group
Manufacturers in this space differentiate themselves through cleanroom manufacturing capability, regulatory expertise, and validated material performance data following gamma sterilization.
Why FMI: https://www.futuremarketinsights.com/why-fmi
Explore More Research Reports by FMI
Refillable, Low-Waste & Modular Packaging Market - https://www.futuremarketinsights.com/reports/refillable-low-waste-modular-packaging-market
Fast Food Containers Market - https://www.futuremarketinsights.com/reports/fast-food-containers-market
White-Label Computer Peripheral Packaging Market - https://www.futuremarketinsights.com/reports/white-label-computer-peripheral-packaging-market
Modular Packaging Architecture for Recyclability Market - https://www.futuremarketinsights.com/reports/modular-packaging-architecture-for-recyclability-market
Wooden Toddler Toy Packaging Market - https://www.futuremarketinsights.com/reports/wooden-toddler-toy-packaging-market
About Future Market Insights (FMI)
Future Market Insights, Inc. (FMI) is an ESOMAR-certified, ISO 9001:2015 market research and consulting organization, trusted by Fortune 500 clients and global enterprises. With operations in the U.S., UK, India, and Dubai, FMI provides data-backed insights and strategic intelligence across 30+ industries and 1200 markets worldwide.
Sudip Saha
Future Market Insights Inc.
+1 347-918-3531
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.